
Nuclear charge: If the nuclear charge ( atomic number) is greater, the electrons are held more tightly by the nucleus and hence the ionization energy will be greater (leading to the mentioned trend 1 within a given period).Electron configuration: This accounts for most elements' IE, as all of their chemical and physical characteristics can be ascertained just by determining their respective electron configuration.The most notable influences that determine ionization energy include: For example, the first three ionization energies are defined as follows:ġst ionization energy is the energy that enables the reaction X ⟶ X + + e − 2nd ionization energy is the energy that enables the reaction X + ⟶ X 2+ + e − 3rd ionization energy is the energy that enables the reaction X 2+ ⟶ X 3+ + e − The nth ionization energy refers to the amount of energy required to remove the most loosely bound electron from the species having a positive charge of ( n − 1). The latter trend results from the outer electron shell being progressively farther from the nucleus, with the addition of one inner shell per row as one moves down the column. Ionization energy generally decreases from top to bottom in a given group (that is, column).Ionization energy generally increases from left to right within a given period (that is, row). 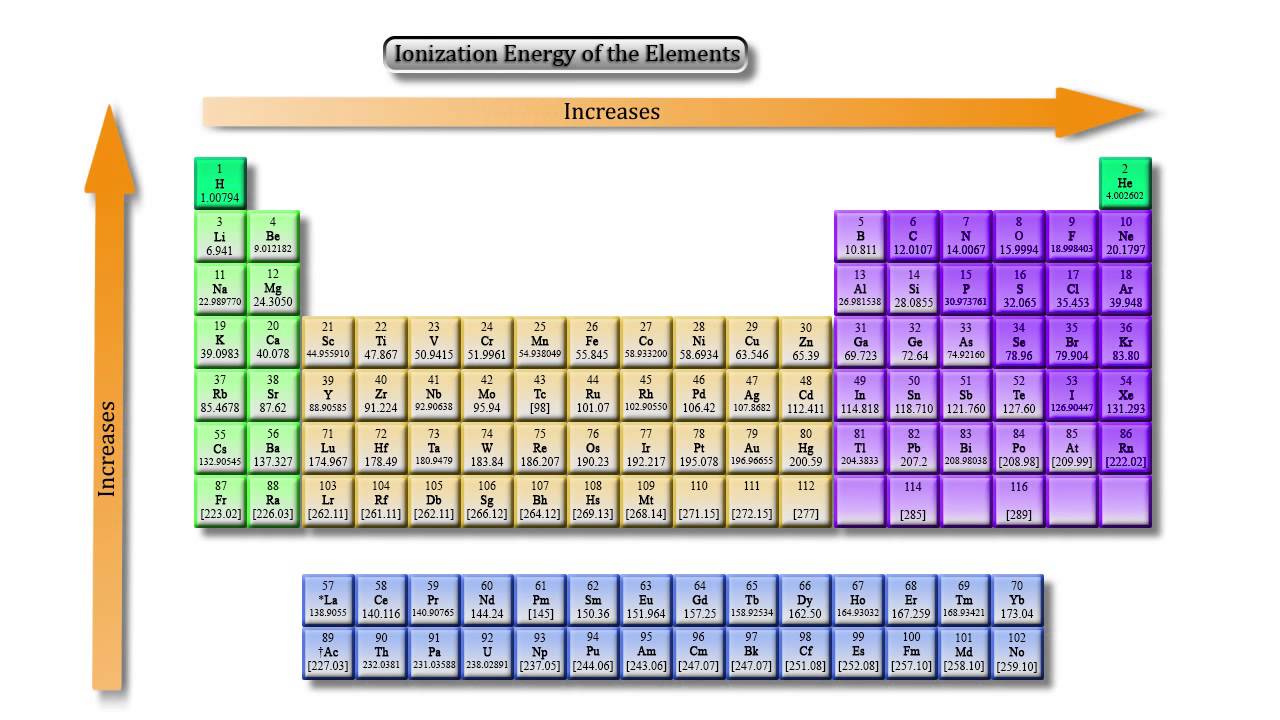
Ĭomparison of ionization energies of atoms in the periodic table reveals two periodic trends which follow the rules of Coulombic attraction: In chemistry, it is expressed as the energy to ionize a mole of atoms or molecules, usually as kilojoules per mole (kJ/mol) or kilocalories per mole (kcal/mol). In physics, ionization energy is usually expressed in electronvolts (eV) or joules (J). Roughly speaking, the closer the outermost electrons are to the nucleus of the atom, the higher the atom's ionization energy. Ionization energy is positive for neutral atoms, meaning that the ionization is an endothermic process. Where X is any atom or molecule, X + is the resultant ion when the original atom was stripped of a single electron, and e − is the removed electron. The first ionization energy is quantitatively expressed as In physics and chemistry, ionization energy ( IE) ( American English spelling), ionisation energy ( British English spelling) is the minimum energy required to remove the most loosely bound electron of an isolated gaseous atom, positive ion, or molecule. Predicted values are used for elements beyond 104. The maximum ionization energy also decreases from the first to the last row in a given column, due to the increasing distance of the valence electron shell from the nucleus. The ionization energy gradually increases from the alkali metals to the noble gases. Ionization energy trends plotted against the atomic number, in units eV.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
